Healthcare organisations generate more data than almost any other industry. The problem is not the data. It is the gap between the data and the insight.
Electronic health records, imaging studies, lab results, claims data, genomic profiles, and remote monitoring streams accumulate at a scale that no human team can manually process with the speed clinical decisions require. Traditional machine learning can close that gap, but building accurate models has historically required specialised data science expertise that most health systems and life sciences firms simply do not have on staff.
Automated machine learning, or AutoML, changes that equation. It does not replace clinical judgment. What it does is make the machinery of predictive analytics in healthcare accessible to the people closest to the clinical problem. This guide explains what AutoML is, how it works in a healthcare context, where it adds measurable value, and what leaders should look for before adopting it.
$2.59B
AutoML global market value in 2025
41.96%
CAGR projected through 2031
What Is AutoML?
AutoML stands for automated machine learning. It refers to software that automates the most time-intensive steps in building a predictive model: selecting the right algorithm, engineering features from raw data, and tuning the model’s internal parameters for optimal accuracy. Steps that once took a team of data scientists weeks can be completed in hours.
Crucially, AutoML does not produce a magic black box. A well-designed platform makes the process transparent and auditable. Most enterprise AutoML tools include explainability modules that show which variables drove a prediction and by how much. This matters enormously in healthcare, where regulators and ethics committees expect clear answers about why an algorithm flagged a patient or recommended a clinical pathway.
The broader shift toward no-code machine learning and AI model automation means that domain experts such as clinical informaticists, quality analysts, and operations leaders can participate meaningfully in building predictive models, rather than waiting for centralised data science teams to prioritise their requests.
How Does AutoML Work?
An AutoML workflow moves through three core stages:
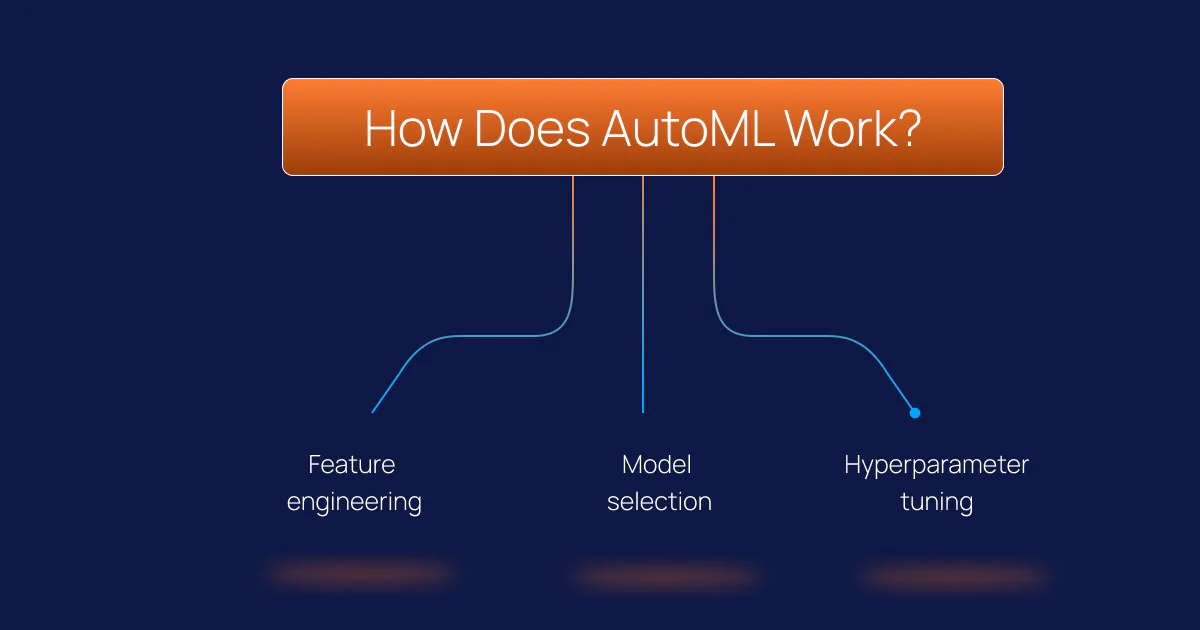
Feature engineering
Raw healthcare data – diagnosis codes, lab values, admission timestamps, medication lists – is transformed into numerical signals a model can use. AutoML platforms identify which transformations produce the most predictive features without manual trial and error. For structured EHR data, this stage often surfaces non-obvious signal combinations that manual feature engineering would miss entirely.
Model selection
The platform tests multiple algorithm families simultaneously, such as gradient boosting, random forests, and neural architectures, and identifies which performs best for the specific data and target outcome. This eliminates the guesswork and hours of experimentation that traditional data science workflows require.
Hyperparameter tuning
Each algorithm has internal settings that control its behaviour. AutoML systematically explores combinations of these settings and converges on a configuration that maximises predictive accuracy without overfitting the training data.
The result is a validated, deployable model built in a fraction of the time. The no-code and low-code interfaces of modern AutoML platforms mean that healthcare teams can initiate model training automation projects independently, review outputs, and iterate based on clinical feedback rather than queuing requests to a centralised data team.
AutoML Use Cases in Healthcare: Where It Matters
The following use cases represent areas where AutoML in healthcare has moved from pilot to production across health systems and life sciences organisations.
Patient Risk Stratification and Readmission Prediction
Unplanned readmissions cost the US healthcare system billions of dollars annually and remain one of the most closely watched quality metrics under CMS value-based care programmes. Machine learning models built on EHR data can predict 30-day readmission risk and in-hospital mortality with AUROC scores reaching 0.93 to 0.94 in large multi-site clinical cohorts. AutoML makes this type of modelling repeatable across facilities without requiring a dedicated data science team at every site.
Chronic Disease Detection and Early Intervention
Cardiovascular risk, diabetes progression, COPD exacerbation risk, and chronic kidney disease staging are all conditions where early prediction enables timely intervention. AutoML frameworks have been applied to coronary artery disease prediction with results demonstrating clinical-grade accuracy; when integrated with SHAP, it improves the explainability and transparency of ML models. Explainable AI in healthcare is not optional; a model that clinicians cannot interrogate will not be adopted regardless of its accuracy scores.
HEDIS and Quality of Care Analytics
Health plans operating under HEDIS and CMS STAR rating frameworks process millions of member records to identify care gaps, track chronic condition management, and optimise quality scores. Automated ML model training accelerates the cycle from data ingestion to population-level insight, enabling health plans to act on gap-in-care signals before the measurement year closes rather than reacting after the fact.
Adverse Event Detection in Pharma
Under 21 CFR Part 11 and FDA pharmacovigilance requirements, pharmaceutical companies must classify and report adverse events from clinical trials and post-market surveillance. AutoML-powered NLP pipelines can process unstructured safety reports, classify event severity, and flag regulatory submission deadlines automatically, reducing the manual burden on safety operations teams while improving reporting consistency.
Clinical Trial Patient Matching
Identifying eligible patients for clinical trials is one of the most expensive and time-consuming stages of pharmaceutical R&D. AI-driven patient matching using AutoML applied to EHR data, genomic profiles, and SNOMED CT-coded diagnoses can accelerate enrolment by narrowing a population of millions to a targeted cohort. By automating the identification of highly specific patient cohorts, AI-driven analytics can compress the clinical recruitment phase – a traditional bottleneck in drug development. In documented industry cases, integrating these automated workflows has helped reduce key stages of the drug discovery and trial lifecycle from a typical 5 to 6-year window down to approximately one year.
The Intersection of AutoML and Large Language Models (LLMs)
| Dimension | AWS | Azure |
|---|---|---|
| BAA mechanism | Signed via AWS Artifact for designated HIPAA accounts | Auto-included in Microsoft Product Terms for qualifying customers |
| HIPAA-eligible services | 166+ services across compute, storage, AI, analytics | Service-level eligibility, validated per workload in Product Terms |
| Native healthcare data layer | Amazon HealthLake (managed FHIR R4 + medical NLP) | Azure Health Data Services (FHIR + DICOM + MedTech in one workspace) |
| Analytics engine | Athena, Redshift, EMR, SageMaker, QuickSight | Synapse Analytics, Databricks, Azure ML, Power BI |
| Identity backbone | AWS IAM, Identity Center, KMS | Microsoft Entra ID, Conditional Access, Azure Key Vault |
| Federal healthcare | AWS GovCloud (US), FedRAMP High | Azure Government, FedRAMP High, IL5 |
| Best fit for | Greenfield FHIR-first analytics, custom ML pipelines, federal health agencies | Microsoft-shop hospitals, imaging-heavy workloads, integrated BI on existing M365 estates |
While AutoML excels at finding patterns in structured data (like lab values and claims), Large Language Models (LLMs) like Med-PaLM 2 or GPT-4o have redefined how we handle unstructured clinical text. In 2026, the most effective healthcare AI strategies don’t choose between the two – they integrate them.
Structured Prediction vs. Narrative Understanding
The core difference lies in the data type. AutoML is your engine for predictive analytics in healthcare, turning EHR tables into risk scores. LLMs, conversely, act as the “clinical interpreter,” summarizing decades of physician notes or extracting SNOMED CT codes from messy discharge summaries.
Are LLMs Trustworthy for Clinical Decisions?
A common question among data leaders is: Can an LLM help with complex clinical decision-making? The answer is “yes, but with guardrails.” While LLMs excel at medical knowledge benchmarks, they can “hallucinate” or miss critical clinical nuances (like the difference between “suspected pneumonia” and a confirmed diagnosis).
To make a healthcare LLM clinically useful and trustworthy, it must be paired with:
- Retrieval-Augmented Generation (RAG): Grounding the LLM in your organization’s specific clinical protocols.
- AutoML Pipelines: Using AutoML to validate the numerical "sanity" of an LLM’s suggestion.
Can Patients Use LLMs Safely?
Patients often ask if they can safely use AI for personal health advice. While LLMs are powerful research tools, they lack the real-time diagnostic accountability of a clinician. In a regulated setting, LLMs are best used to assist doctors – reducing administrative burnout and identifying eligible patients for clinical trials – rather than replacing human clinical judgment.
AutoML vs. Traditional Machine Learning: The Practical Difference
| Traditional Machine Learning | AutoML |
|---|---|
| Requires specialised data science expertise | Accessible to domain experts and business analysts |
| Model selection is manual and iterative | Automated model selection across multiple algorithm families |
| Feature engineering is labour-intensive | Automated feature transformation and selection |
| Deployment timelines measured in weeks to months | Model training automation reduces timelines to hours or days |
| Explainability depends on team capability | Built-in explainability (SHAP, LIME) as standard in enterprise platforms |
| High cost per model at scale | Lower cost per model, enabling broader deployment across use cases |
What Makes a Healthcare AutoML Trustworthy?
Healthcare data science operates under constraints that most other industries do not face. Before selecting an AutoML platform or a clinical machine learning services partner, IT and data leaders should consider the following aspects:
- Explainability:The platform must generate human-interpretable explanations for every prediction, not just performance scores. SHAP values and similar attribution methods allow clinicians to verify that a model is drawing on clinically plausible signals, not spurious correlations in the training data.
- Regulatory alignment:Deployments in healthcare must be consistent with HIPAA, and in pharma and life sciences contexts, with 21 CFR Part 11 and GxP standards. Data governance, audit trails, and access controls are operational requirements, not optional features.
- Bias detection and model monitoring:Models trained on historical EHR data can encode historical disparities across demographic groups. Trustworthy platforms include fairness checks and ongoing monitoring to detect data drift over time and trigger retraining when model performance degrades.
- Validation on representative populations:A model validated on one hospital system's patient population does not automatically generalise to another. Clinical machine learning platforms should support multi-site validation and transfer learning to minimise this risk before production deployment.
- PhD-level clinical domain expertise in the loop:Automation handles the computational burden, but clinical and scientific domain knowledge must shape the problem definition, feature selection, and outcomes assessment. AutoML without clinical oversight produces technically functional but clinically uninformed models.
How Intuceo Integrates AutoML in Healthcare
Intuceo is a PhD-led AI, ML, and data analytics consulting firm specialising in regulated industries. Its proprietary AutoML accelerators, part of the Intuceo-Ax platform, are purpose-built for healthcare and life sciences environments where explainability, compliance, and clinical precision are operational requirements.
- Predict Engine: Forecasting models for patient risk stratification, HEDIS gap analysis, and Potentially Preventable Event (PPE) reduction, deployed against Epic and Cerner EHR data.
- Text Analytics Engine (NLP): Automated adverse event classification for pharma safety operations, aligned with 21 CFR Part 11 reporting requirements.
- AgentCare AI: An agentic AI solution for clinical trial patient matching that maps EHR profiles to eligibility criteria at scale using SNOMED CT entity resolution.
- 360° Patient Insights (Intuceo-Ix): Unified intelligence across EHRs, social determinants of health (SDoH), and home care records, creating a single patient view for care management teams.
Every engagement is governed by Intuceo's iPDLC methodology, ensuring that clinical domain expertise drives problem framing and outcome evaluation, not just engineering velocity.
Frequently Asked Questions
1.Can AutoML replace a data scientist in healthcare?
AutoML automates the most repetitive and computationally intensive parts of building a predictive model, but it does not replace the clinical domain expertise needed to define the right problem, identify the right data sources, and evaluate whether a model’s predictions make clinical sense. In practice, AutoML shifts data scientists toward higher-value work: problem framing, clinical validation, and deployment oversight.
2.What is explainable AI in healthcare and why does it matter?
Explainable AI refers to methods that make a model’s predictions interpretable to a human reviewer. In healthcare, this means a clinician or compliance officer can see which patient variables contributed most to a risk score and to what degree. Without explainability, clinicians have no basis for trusting or appropriately challenging a model’s output. Regulatory bodies including the FDA have signalled increasing expectations around algorithm transparency for software as a medical device (SaMD).
3.What data does AutoML need in a healthcare context?
AutoML models in healthcare most commonly draw on structured EHR data (diagnosis codes, procedure codes, lab results, medications, vital signs), administrative data (claims, encounter history, admission and discharge records), and where available, genomic or imaging data. The quality, consistency, and completeness of that data determines the ceiling on model performance. Organisations with strong data governance and standardised EHR adoption typically see faster time-to-production on clinical machine learning projects.
4.Is AutoML compliant with HIPAA?
AutoML platforms themselves are not inherently HIPAA-compliant. Compliance depends on how the platform is deployed, how protected health information (PHI) is accessed and stored, and whether appropriate business associate agreements are in place. Healthcare organisations should evaluate vendor security architecture, data residency options, and audit logging capabilities as part of any AutoML procurement or services engagement.
5.What is the difference between AutoML and no-code AI?
AutoML specifically refers to automation of the machine learning model-building process: feature engineering, algorithm selection, and hyperparameter tuning. No-code AI is a broader category covering tools that allow users to build AI-powered applications through visual interfaces without programming. Many AutoML platforms include no-code interfaces, but not all no-code AI tools include full AutoML functionality.

 Artificial Intelligence
Artificial Intelligence DataOps & Engineering
DataOps & Engineering Digital Engineering
Digital Engineering Enterprise Transformation
Enterprise Transformation Healthcare
Healthcare Advanced Manufacturing
Advanced Manufacturing Supply Chain & Transportation
Supply Chain & Transportation Engineering & Auto
Engineering & Auto Public Sector & Strategic Markets
Public Sector & Strategic Markets iPDLC (Lifecycle)
iPDLC (Lifecycle) iTMS
iTMS Modular AI
Modular AI Blogs
Blogs Technical Briefs
Technical Briefs Use Cases
Use Cases